OU NanoLab/NSF NUE/Bumm & Johnson
The Kelvin Effect
Atoms
of liquid on the surface of a small droplet are held less tightly compared to atoms on a flat (bulk) liquid surface. High curvatures effectively reduce the coordination number of the
surface atoms making them easier
to evaporate. Thus the liquid has a
higher vapor pressure over small
liquid droplets compared to bulk liquid.
The effect of curvature on the
vapor pressure of liquids is the Kelvin effect.
Positive curvature:
liquid in drops has a higher vapor pressure that bulk.
Negative curvature:
liquid in pores has a lower vapor pressure than bulk.
The vapor pressure P relative to the bulk P0 can be found using the Kelvin equation, show here for spherical surfaces of radius r.
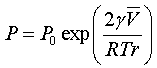

The other parameters are:
γ the surface tension,
the molar volume of the liquid,
R the gas constant, and
T the absolute temperature.
γ the surface tension,
the molar volume of the liquid,
R the gas constant, and
T the absolute temperature.