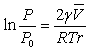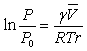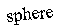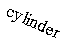OU NanoLab/NSF NUE/Bumm & Johnson
The Kelvin Equation
The surface tension causes an increased chemical
potential for a molecule inside
a droplet. This is manifested as an
increase in the vapor pressure P of the liquid droplet compared to that of the bulk
liquid P0. The is described
by the Kelvin equation. Two radii of
curvature appear in the result,
r1 and r2. For a sphere both terms are equal, but for a
cylindrical surface one
term vanishes because one radius is infinite (flat).
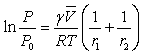

The other parameters are:
γ the surface tension,
the molar volume of the liquid,
R the gas constant, and
T the absolute temperature.
γ the surface tension,
the molar volume of the liquid,
R the gas constant, and
T the absolute temperature.